Welcome to the award-winning BU Research Blog!
Our research shapes and changes the world around us, providing solutions to real-world problems and informing the education we deliver.
Click here to view the full blog, or browse the categories below and with the menu above.
BU staff can login below:
Don’t miss a post!
Subscribe for the BU Research Digest, delivered freshly every day.
Funding opportunities
View current funding opportunities »- ESRC Festival of Social Science 2026: Application Deadline Extended to Thursday 25 June 2026
- Apply now to take part in the 2026 ESRC Festival of Social Science
- Reminder: Register for the ESRC Festival of Social Science 2026 Information Session
- ECR Funding Open Call: Research Culture & Community Grant – Apply now
- FLF UKRI round 11- internal launch and timelines
- ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December
BU research
 Expressions of Interest invited for Research Ethics Roles starting in 2026/27Interested in getting an insight into the nuts-and-bolts of how research is done across the university? Want to help make... Read more »
Expressions of Interest invited for Research Ethics Roles starting in 2026/27Interested in getting an insight into the nuts-and-bolts of how research is done across the university? Want to help make... Read more » Expressions of Interest invited from senior academics to join the BU REF Appeals Panel Our BU REF 2029 Code of Practice states that in the event of an appeal, the Deputy Vice-Chancellor will convene and chair the BU REF Appeals Panel to... Read more »
Expressions of Interest invited from senior academics to join the BU REF Appeals Panel Our BU REF 2029 Code of Practice states that in the event of an appeal, the Deputy Vice-Chancellor will convene and chair the BU REF Appeals Panel to... Read more » REF mock exercise 2026 – nominate your research outputs on BRIANThe next Research Excellence Framework (REF) mock exercise opens today. Following the success of our REF21 submission, we have ambitious... Read more »
REF mock exercise 2026 – nominate your research outputs on BRIANThe next Research Excellence Framework (REF) mock exercise opens today. Following the success of our REF21 submission, we have ambitious... Read more » ESRC Festival of Social Science 2026: Application Deadline Extended to Thursday 25 June 2026The 24th annual ESRC Festival of Social Science will take place from Saturday 17 October to Saturday 7 November 2026, with... Read more »
ESRC Festival of Social Science 2026: Application Deadline Extended to Thursday 25 June 2026The 24th annual ESRC Festival of Social Science will take place from Saturday 17 October to Saturday 7 November 2026, with... Read more » First publication for two CMWH PhD studentsCongratulations to CMWH doctoral student Jennah Evans who has published the first paper from her PhD in the Journal of... Read more »
First publication for two CMWH PhD studentsCongratulations to CMWH doctoral student Jennah Evans who has published the first paper from her PhD in the Journal of... Read more » Another CMWH publicationCongratulations to CMWH doctoral student Jennah Evans who has published the first paper from her PhD in the Journal of... Read more »
Another CMWH publicationCongratulations to CMWH doctoral student Jennah Evans who has published the first paper from her PhD in the Journal of... Read more »
international
PG research
Research lifecycle
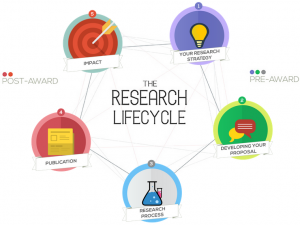 Our Research Lifecycle diagram is a jazzy interactive part of the BU Research Blog that shows the support and initiatives that are available to staff and students at each stage of the research lifecycle.
Our Research Lifecycle diagram is a jazzy interactive part of the BU Research Blog that shows the support and initiatives that are available to staff and students at each stage of the research lifecycle.











