Welcome to the award-winning BU Research Blog!
Our research shapes and changes the world around us, providing solutions to real-world problems and informing the education we deliver.
Click here to view the full blog, or browse the categories below and with the menu above.
BU staff can login below:
Don’t miss a post!
Subscribe for the BU Research Digest, delivered freshly every day.
Funding opportunities
View current funding opportunities »- Horizon Europe Cluster 3 (Civil Security for Society) 2026 Calls Now Open
- MSCA Doctoral Networks 2026 Call Information Webinar
- ESRC Festival of Social Science 2026: Application Deadline Extended to Thursday 25 June 2026
- Apply now to take part in the 2026 ESRC Festival of Social Science
- Reminder: Register for the ESRC Festival of Social Science 2026 Information Session
- ECR Funding Open Call: Research Culture & Community Grant – Apply now
BU research
 Official book launch at Bournemouth UniversityLast night Bournemouth University hosted the official launch at of the book Early Labour and Maternity Care: Research for Practice published by... Read more »
Official book launch at Bournemouth UniversityLast night Bournemouth University hosted the official launch at of the book Early Labour and Maternity Care: Research for Practice published by... Read more » Take a Break: Join the Creative Wellbeing EventSupported by the ECR Research Culture and Community Grant, this upcoming interactive session offers BU researchers a dedicated space to... Read more »
Take a Break: Join the Creative Wellbeing EventSupported by the ECR Research Culture and Community Grant, this upcoming interactive session offers BU researchers a dedicated space to... Read more » Strengthening BU’s Research Culture: A Look Inside the Mixed Methods WorkshopBridging the Methodological Divide: ECR Led Workshop Explores the Power of Mixed Methods Research On Wednesday 6 May 2026, Early... Read more »
Strengthening BU’s Research Culture: A Look Inside the Mixed Methods WorkshopBridging the Methodological Divide: ECR Led Workshop Explores the Power of Mixed Methods Research On Wednesday 6 May 2026, Early... Read more » Psychology, Psychiatry and Neuroscience academics – would you like to get more involved in preparing our next REF submission?We are currently recruiting for an Impact Champion to help support preparation for our next REF Submission to UoA4: Psychology,... Read more »
Psychology, Psychiatry and Neuroscience academics – would you like to get more involved in preparing our next REF submission?We are currently recruiting for an Impact Champion to help support preparation for our next REF Submission to UoA4: Psychology,... Read more » Geography and Environmental Studies academics – would you like to get more involved in preparing our next REF submission?We are currently recruiting for an Output Champion and Impact Champion to help support preparation for our next REF Submission to... Read more »
Geography and Environmental Studies academics – would you like to get more involved in preparing our next REF submission?We are currently recruiting for an Output Champion and Impact Champion to help support preparation for our next REF Submission to... Read more » Three Minute Thesis (3MT®) 2026: Register to Support our PGRsFollowing the round of pre-recorded submissions in April, the stage is set for the 2026 BU Three Minute Thesis (3MT®)... Read more »
Three Minute Thesis (3MT®) 2026: Register to Support our PGRsFollowing the round of pre-recorded submissions in April, the stage is set for the 2026 BU Three Minute Thesis (3MT®)... Read more »
international
PG research
Research lifecycle
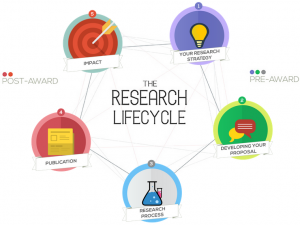 Our Research Lifecycle diagram is a jazzy interactive part of the BU Research Blog that shows the support and initiatives that are available to staff and students at each stage of the research lifecycle.
Our Research Lifecycle diagram is a jazzy interactive part of the BU Research Blog that shows the support and initiatives that are available to staff and students at each stage of the research lifecycle.











