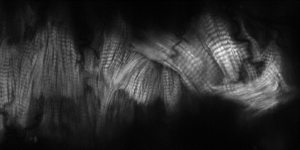
Every beat of the heart is finely tuned to eject a certain amount of blood. As we exercise, more blood flows into the heart, the cardiac muscle stretches and this leads to an increased force of contraction. Known as the Frank-Starling law, it is one of the most important aspects of human cardiac physiology but the molecular mechanisms are not entirely understood.
We do know that increases to the calcium levels in the heart cells (cardiomyocytes) support stronger contractions (anyone remember the ‘sliding ratchet model’ from GCSE biology!?) but how this calcium is regulated by stretch is not fully understood. What my colleagues and I have established (to be published in Frontiers of Physiology) is that a ‘mechanosensitive’ protein known as Piezo helps increase calcium when the cardiomyocytes are stretched. A lot of this work was done at BU’s Drosophila (fruit fly) genetics facility in Dorset House, using physiological tests of heart function in flies without the Piezo protein. When stretched, normal hearts respond by releasing more calcium and they continue to beat. In Piezo mutants, there’s no increase in calcium and the hearts often stop beating.
This is an important observation that contributes to our fundamental understanding of cardiac physiology and points to Piezo as a protein of considerable interest when considering the underlying causes of cardiac dysfunction in disease and ageing.
Paul Hartley.
(The image shows the contractile protein ‘scaffold’ within an insect heart)











 Reminder: Register for the ESRC Festival of Social Science 2026 Information Session
Reminder: Register for the ESRC Festival of Social Science 2026 Information Session Deadline Approaching: Submit your Poster for the Research Conference by Monday 27 April
Deadline Approaching: Submit your Poster for the Research Conference by Monday 27 April BU academics publish in Nepal national newspaper
BU academics publish in Nepal national newspaper New BU Physiology paper
New BU Physiology paper Gender and street names
Gender and street names ECR Funding Open Call: Research Culture & Community Grant – Apply now
ECR Funding Open Call: Research Culture & Community Grant – Apply now ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December
ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December MSCA Postdoctoral Fellowships 2025 Call
MSCA Postdoctoral Fellowships 2025 Call ERC Advanced Grant 2025 Webinar
ERC Advanced Grant 2025 Webinar Update on UKRO services
Update on UKRO services European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease
European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease