
RDS NEWS
NIHR Grant Applications Seminar and Support event
Our popular seminar continues online and will next take place in March 2023. Watch this space for further details.
In the meantime, you can register your interest with us and once details of the event are available, you will receive an email with a weblink to find out more.
Alternatively, you can contact our Coordinating Centre on 01392 726724 or rds.sw@nihr.ac.uk, or your local RDS office to find out more.
NIHR News
Experimental treatments for cancer receive funding boost
The NIHR is contributing £21.6 million to the network of Experimental Cancer Medicine Centres (ECMCs) as part of a £47.5 million funding package over the next 5 years. Find out more
Workshops and Events
NIHR webinar: Discover funding opportunities for qualitative researchers
7 February 2023, 10.30am – 12.30pm. ONLINE
This webinar is hosted by the Qualitative Workstream of the NIHR Methodology Incubator and the NIHR Academy. The webinar aims to support the careers of qualitative and mixed methods researchers by outlining funding opportunities for health and care research. Speakers will provide an overview of funding schemes including personal fellowships, small project grants, large research programmes and grants for methods research. Participants will hear from NIHR Senior Investigators, Directors of funding schemes as well as people who manage schemes and sit on funding committees. You will have an opportunity to ask questions to the speakers in a Q&A session at the end. Find out more.
NIHR webinar: Embedding PPIE in your research
21 February 2023, 1.00pm – 2.00pm. ONLINE
This webinar will demonstrate how to integrate PPIE into your research and focus on working with underserved groups or topics. The difficulty facing research teams, especially early career researchers, is when and how to use the public and patients.
We know that the communities mostly likely to experience health inequalities and benefit most from research are the underserved and underrepresented. The webinar will explore ways of answering the following questions:
- How would you target a group/community to participate in your research?
- What tools, techniques and partners would you enlist to make sure your target community is able to get involved in your research?
- How would you know if you got it right?
Find out more.
NCRM webinar series: Data resources for mental health and wellbeing research
6 March 2023. ONLINE
A two-part webinar series will explain how to access and use datasets on mental health and wellbeing. Organised by the Data Resources Training Network (DRTN), the free series begins on Monday, 6 March with a webinar on secondary, quantitative data. The speakers will be Sally McManus from NatCen, Eoin McElroy from Ulster University and Mark Elliot from The University of Manchester. To complement the series, NCRM has collated a suite of resources relating to mental health and wellbeing, which will be launched to coincide with the events. Find out more.
Funding Opportunities
Funding deadline calendar
For an overview of NIHR calls and ongoing funding opportunities, please see our funding deadlines webpage
Latest NIHR funding calls
Fellowship Programme
Pre-doctoral Fellowship (Round 5)
HEE-NIHR Integrated Clinical and Practitioner Academic Programme
Pre-doctoral Clinical and Practitioner Academic Fellowship (PCAF) Round 6
Health and Social Care Delivery Research (HSDR) Programme
23/8 Care (Education) and Treatment Reviews (C(E)TRs) for people with learning disabilities and/or autistic people
Local Authority Academic Fellowship Programme
Pre-doctoral Local Authority Fellowship (PLAF) Round 3
Public Health Research (PHR) Programme
23/12 Health Determinant Research Collaborations (HDRCs)
Themed call
Compound pressures
Your local branch of the NIHR RDS (Research Design Service) is based within the BU Clinical Research Unit (BUCRU) should you need help with any grant applications. We advise on all aspects of developing an application and can review application drafts as well as put them to a mock funding panel (run by RDS South West) known as Project Review Committee, which is a fantastic opportunity for researchers to obtain a critical review of a proposed grant application before this is sent to a funding body or if you’re hoping to resubmit the panel can provide some excellent tips and feedback.
Contact us as early as possible to benefit fully from the advice
Feel free to call us on 01202 961939 or send us an email.
 This week four postgraduate midwifery students from Bournemouth University attended the Royal College of Midwives annual Education & Research conference in London. Their contributions included studies on: (1) ‘A Unique Approach to Smoking Cessation During Pregnancy’ by Ph.D. student Louise Barton; (2) Investigating how women make decisions about prescribed psychiatric medication use during pregnancy by M.Res. student Jessica Correia; (3) Harnessing midwives’ research delivery expertise to encourage medics’ participation in research’ by M.Res. student Susara Blunden; and (4) ‘Personalised care for women of advanced maternal age, from conception to postnatal care: A mixed-methods study’ by Ph.D. student Joanne Rack. Joanne was also at this conference in her capacity of the newly appointed Editor-in-Chief of The Practising Midwife.
This week four postgraduate midwifery students from Bournemouth University attended the Royal College of Midwives annual Education & Research conference in London. Their contributions included studies on: (1) ‘A Unique Approach to Smoking Cessation During Pregnancy’ by Ph.D. student Louise Barton; (2) Investigating how women make decisions about prescribed psychiatric medication use during pregnancy by M.Res. student Jessica Correia; (3) Harnessing midwives’ research delivery expertise to encourage medics’ participation in research’ by M.Res. student Susara Blunden; and (4) ‘Personalised care for women of advanced maternal age, from conception to postnatal care: A mixed-methods study’ by Ph.D. student Joanne Rack. Joanne was also at this conference in her capacity of the newly appointed Editor-in-Chief of The Practising Midwife.  Congratulations to these postgraduate students and their supervisors.
Congratulations to these postgraduate students and their supervisors.









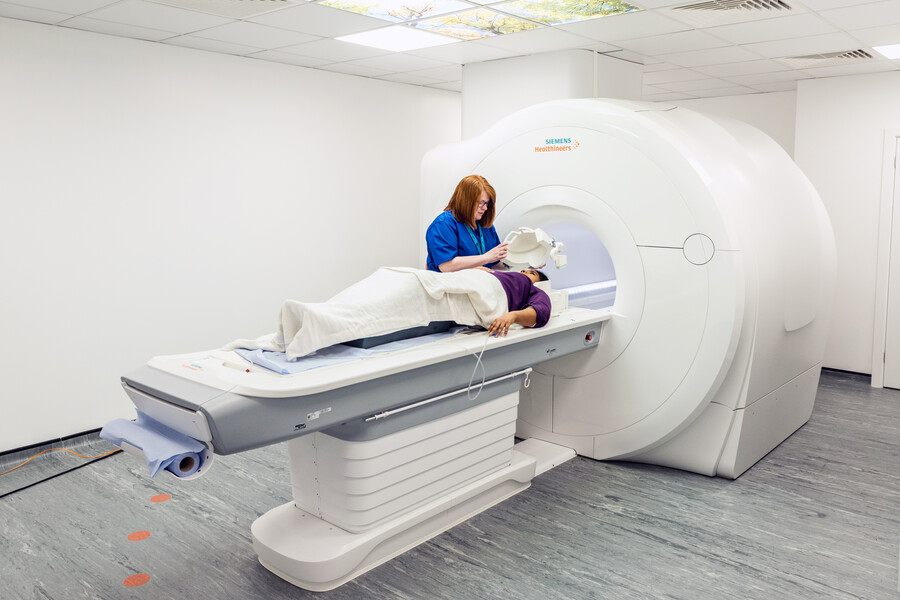 The Institute of Medical Imaging and Visualisation (IMIV) is pleased to announce the launch of the IMIV MRI Research Project Scheme 2023.
The Institute of Medical Imaging and Visualisation (IMIV) is pleased to announce the launch of the IMIV MRI Research Project Scheme 2023. Applications close on Friday 7th July 2023.
Applications close on Friday 7th July 2023. 
















 Official book launch at Bournemouth University
Official book launch at Bournemouth University Take a Break: Join the Creative Wellbeing Event
Take a Break: Join the Creative Wellbeing Event Psychology, Psychiatry and Neuroscience academics – would you like to get more involved in preparing our next REF submission?
Psychology, Psychiatry and Neuroscience academics – would you like to get more involved in preparing our next REF submission? Three Minute Thesis (3MT®) 2026: Register to Support our PGRs
Three Minute Thesis (3MT®) 2026: Register to Support our PGRs Horizon Europe Cluster 3 (Civil Security for Society) 2026 Calls Now Open
Horizon Europe Cluster 3 (Civil Security for Society) 2026 Calls Now Open MSCA Doctoral Networks 2026 Call Information Webinar
MSCA Doctoral Networks 2026 Call Information Webinar ESRC Festival of Social Science 2026: Application Deadline Extended to Thursday 25 June 2026
ESRC Festival of Social Science 2026: Application Deadline Extended to Thursday 25 June 2026 Reminder: Register for the ESRC Festival of Social Science 2026 Information Session
Reminder: Register for the ESRC Festival of Social Science 2026 Information Session ECR Funding Open Call: Research Culture & Community Grant – Apply now
ECR Funding Open Call: Research Culture & Community Grant – Apply now ERC Advanced Grant 2025 Webinar
ERC Advanced Grant 2025 Webinar Update on UKRO services
Update on UKRO services European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease
European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease