 The Department of Design and Engineering at Bournemouth University has a reputation for its Human-Centred Design (HCD) work. In our interdisciplinary Sonamoni project we have HCD at its centre. The Sonamoni project is coordinated by Bournemouth University in collaboration with the University of the West of England (Bristol), the University of Southampton, and the Royal National Lifeboat Institution (RNLI), Design Without Border (DWB) in Uganda and Centre for Injury Prevention and Research, Bangladesh (CIPRB). The interdisciplinary team at Bournemouth University covers three faculties and six academics: Dr. Mavis Bengtsson, Dr. Kyungjoo Cha, Dr. Mehdi Chowdhury, Dr. Yong Hun Lim, Mr. John Powell, and Prof. Edwin van Teijlingen.
The Department of Design and Engineering at Bournemouth University has a reputation for its Human-Centred Design (HCD) work. In our interdisciplinary Sonamoni project we have HCD at its centre. The Sonamoni project is coordinated by Bournemouth University in collaboration with the University of the West of England (Bristol), the University of Southampton, and the Royal National Lifeboat Institution (RNLI), Design Without Border (DWB) in Uganda and Centre for Injury Prevention and Research, Bangladesh (CIPRB). The interdisciplinary team at Bournemouth University covers three faculties and six academics: Dr. Mavis Bengtsson, Dr. Kyungjoo Cha, Dr. Mehdi Chowdhury, Dr. Yong Hun Lim, Mr. John Powell, and Prof. Edwin van Teijlingen.
Last month two staff from CIPRB, Notan Chandra Dutta and Mirza Shibat Rowshan visited DBW in Uganda, as part of so-called South-South learning. Their objective was to share (1) knowledge and experience of using HCD techniques and (2) best practices of drowning prevention in both countries. Utilizing HCD techniques, Sonamoni is working to identify and prioritize potential solutions, develop prototypes, and assess the acceptability of the interventions to reduce drowning deaths among old children under two in Bangladesh.
During the visit, Notan and Shibat participated a four-day ideation workshop with the fisher community near Lake Victoria, organized by DWB. In the workshop, different HCD tools were used along with other group activities to generate and refine ideas for the solutions. The generated ideas were recorded by visualization tools. Notan and Shibat also attended a session on the principles of creative facilitation of HCD, including the need to understand the problem, role of the facilitator and other stakeholders. Various visualization tools were discussed, e.g. ‘journey maps’, ‘stakeholder map’, ‘context map’ and different types of sketches. Notan shared CIPRB’s experiences of managing the best drowning prevention practices and its challenges from Bangladesh context.
 This international project funded by the National Institute for Health and Care Research (NIHR) through their Research and Innovation for Global Health Transformation programme, also includes a BU-based PhD student, Mr. Md. Shafkat Hossein. Last week Shafkat presented our Sonamoni project in lecture to BU Engineering students at Talbot campus.
This international project funded by the National Institute for Health and Care Research (NIHR) through their Research and Innovation for Global Health Transformation programme, also includes a BU-based PhD student, Mr. Md. Shafkat Hossein. Last week Shafkat presented our Sonamoni project in lecture to BU Engineering students at Talbot campus.
Prof. Edwin van Teijlingen
Centre for Midwifery & Women’s Health
 We are currently recruiting to the INSIGHT MRes Programme for September 2026. These fully funded M.Res. studentships are available to nurses, midwives and allied health rpofessionals as well as final year nursing, midwifery and allied health students working towards professional registration in the UK. More information can be found here!
We are currently recruiting to the INSIGHT MRes Programme for September 2026. These fully funded M.Res. studentships are available to nurses, midwives and allied health rpofessionals as well as final year nursing, midwifery and allied health students working towards professional registration in the UK. More information can be found here!


















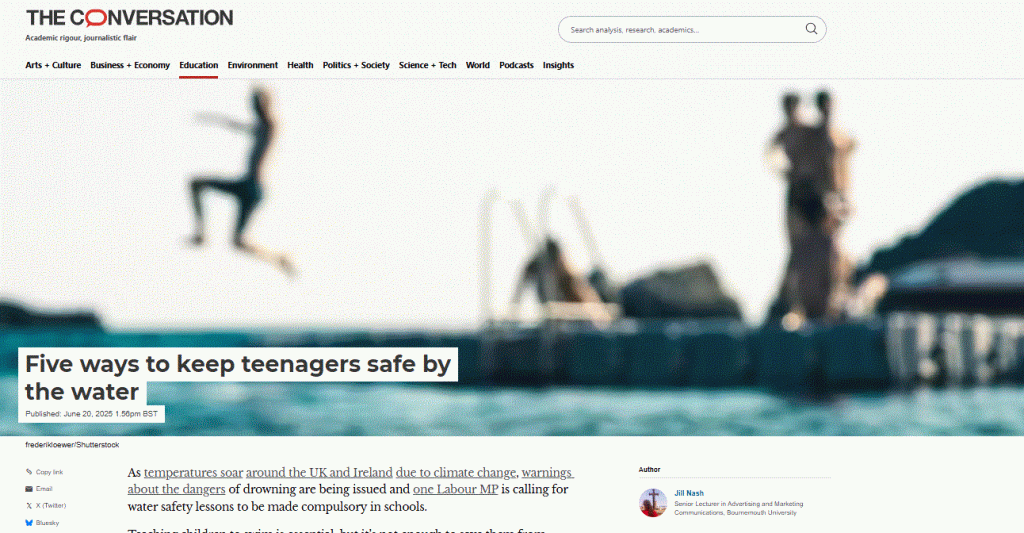



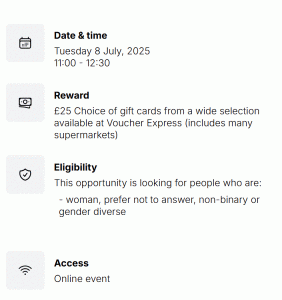

























 Apply now to take part in the 2026 ESRC Festival of Social Science
Apply now to take part in the 2026 ESRC Festival of Social Science Reminder: Register for the ESRC Festival of Social Science 2026 Information Session
Reminder: Register for the ESRC Festival of Social Science 2026 Information Session Deadline Approaching: Submit your Poster for the Research Conference by Monday 27 April
Deadline Approaching: Submit your Poster for the Research Conference by Monday 27 April BU academics publish in Nepal national newspaper
BU academics publish in Nepal national newspaper New BU Physiology paper
New BU Physiology paper ECR Funding Open Call: Research Culture & Community Grant – Apply now
ECR Funding Open Call: Research Culture & Community Grant – Apply now ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December
ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December MSCA Postdoctoral Fellowships 2025 Call
MSCA Postdoctoral Fellowships 2025 Call ERC Advanced Grant 2025 Webinar
ERC Advanced Grant 2025 Webinar Update on UKRO services
Update on UKRO services European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease
European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease
Explore our work, meet our partners, and find out how you can collaborate with us by clicking here! MIHERC is led by Sheffield Hallam University, with Bournemouth University as a key partner and the important funding coming from NIHR (National Institute for Health and Care Research) Maternity Challenge Initiative. The BU key academics are: Huseyin Dogan, Vanora Hundley, Edwin van Teijlingen, and Deniz Çetinkaya. Please share with all who may be interested.