 In the last month we had several FHSS-Psychology success stories. The first one was a recently accepted joint publication between Mr. Paul Fairbairn and Dr. Fotini Tsofliou in the Department of Rehabilitation and Sport Sciences, Dr. Andrew Johnson in BU’s Department of Psychology. The joint paper is called ‘Effects of a high DHA multi-nutrient supplement and exercise on mobility and cognition in older women (MOBILE): A randomised semi-blinded placebo controlled study” in the British Journal of Nutrition [1].
In the last month we had several FHSS-Psychology success stories. The first one was a recently accepted joint publication between Mr. Paul Fairbairn and Dr. Fotini Tsofliou in the Department of Rehabilitation and Sport Sciences, Dr. Andrew Johnson in BU’s Department of Psychology. The joint paper is called ‘Effects of a high DHA multi-nutrient supplement and exercise on mobility and cognition in older women (MOBILE): A randomised semi-blinded placebo controlled study” in the British Journal of Nutrition [1].
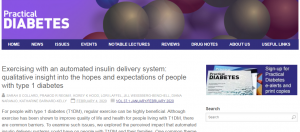
Secondly, Dr. Sarah Collard in the Department of Psychology, Dr. Pramod Regmi in the Department of Nursing Science and FHSS Visiting Professor Katherine Barnard-Kelly are to be congratulated on their publication: ‘Exercising with an automated insulin delivery system: qualitative insight into the hopes and expectations of people with type 1 diabetes’ [2].
 And last, but not least, Dr. Bibha Simkhada in the Department of Nursing Science together with FHSS colleagues Dr. Michele Board and Prof. Edwin van Teijlingen and Dr. Shanti Shanker in the Department of Psychology were awarded £17,180 in the most recent internal GCRF call. Their proposed project ‘The key issues in Dementia in South Asia’ will run from 2020-2021. Both Dr. Simkhada and Dr. Shanker are Global Engagement Lead (GEL) in their respective departments.
And last, but not least, Dr. Bibha Simkhada in the Department of Nursing Science together with FHSS colleagues Dr. Michele Board and Prof. Edwin van Teijlingen and Dr. Shanti Shanker in the Department of Psychology were awarded £17,180 in the most recent internal GCRF call. Their proposed project ‘The key issues in Dementia in South Asia’ will run from 2020-2021. Both Dr. Simkhada and Dr. Shanker are Global Engagement Lead (GEL) in their respective departments.
Good to see so many great cross-BU collaborations!
Professor Edwin van Teijlingen
CMMPH
References:
Fairbairn, P., Tsofliou, F., Johnson, A., Dyall, S.C. (2010) Effects of a high DHA multi-nutrient supplement and exercise on mobility and cognition in older women (MOBILE): A randomised semi-blinded placebo controlled study, British Journal of Nutrition (accepted).
Collard, S.S., Regmi, P.R., Hood, K.K., Laffel, L., Weissberg-Benchell, J., Naranjo, D., Barnard-Kelly, K. (2020) Exercising with an automated insulin delivery system: qualitative insight into the hopes and expectations of people with type 1 diabetes, Practical Diabetes 2020; 37(1): 19–23.





 healthy adults. This is the biggest study of its kind and those who volunteer for this research study will get the chance to see their spine move in real-time.
healthy adults. This is the biggest study of its kind and those who volunteer for this research study will get the chance to see their spine move in real-time.















 New BU Physiology paper
New BU Physiology paper Gender and street names
Gender and street names Help Shape the Future of Research at BU: Postgraduate Research Experience Survey 2026 Now Open
Help Shape the Future of Research at BU: Postgraduate Research Experience Survey 2026 Now Open 3C Event: Research Culture, Community & Cherry Blossom – Tuesday 14 April
3C Event: Research Culture, Community & Cherry Blossom – Tuesday 14 April REMINDER: 3MT® Competition – Deadline 9am Monday 20 April
REMINDER: 3MT® Competition – Deadline 9am Monday 20 April New academic paper on Nepal
New academic paper on Nepal ECR Funding Open Call: Research Culture & Community Grant – Apply now
ECR Funding Open Call: Research Culture & Community Grant – Apply now ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December
ECR Funding Open Call: Research Culture & Community Grant – Application Deadline Friday 12 December MSCA Postdoctoral Fellowships 2025 Call
MSCA Postdoctoral Fellowships 2025 Call ERC Advanced Grant 2025 Webinar
ERC Advanced Grant 2025 Webinar Update on UKRO services
Update on UKRO services European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease
European research project exploring use of ‘virtual twins’ to better manage metabolic associated fatty liver disease